Ethamsylate IP
Price 500 INR/ Kilograms
Ethamsylate IP Specification
- Properties
- Hemostatic agent, promotes capillary endothelial stability, reduces bleeding time
- Smell
- Odorless
- Molecular Weight
- 279.31 g/mol
- Ingredients
- Ethamsylate IP
- CAS No
- 2624-44-4
- Taste
- Characteristic
- Ph Level
- Neutral (Approx. 7)
- Physical Form
- Powder
- Storage
- Store in tightly closed containers at room temperature, protected from moisture and light
- Poisonous
- Non-poisonous
- Molecular Formula
- C10H17NO5S
- Product Type
- Pharmaceutical Raw Material
- Appearance
- White to off-white powder
- Shape
- Crystalline Powder
- Usage
- Active pharmaceutical ingredient for formulation of tablets, capsules, injectables
- Purity
- >99%
- HS Code
- 29420090
- Grade
- IP (Indian Pharmacopoeia), Pharmaceutical Grade
- EINECS No
- 220-098-7
- Shelf Life
- 5 years
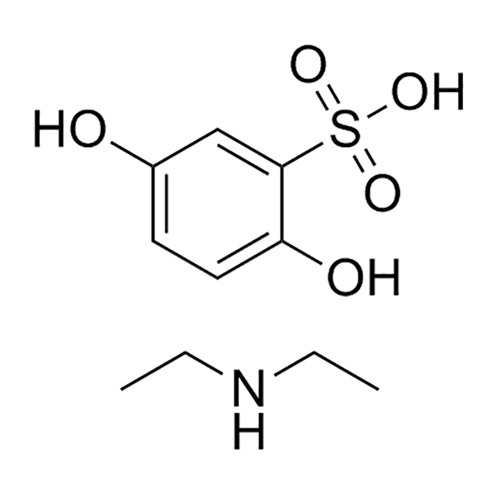
- Structural Formula
- Available on request
- Solubility
- Soluble in water
- Melting Point
- 126-131C
- Application
- Used in the treatment of hemorrhage, capillary bleeding, and prevention of post-operative bleeding
- Density
- 1.35 Gram per cubic centimeter(g/cm3)
- Heavy Metals
- Not more than 0.002%
- Quality Standards
- Indian Pharmacopoeia (IP), GMP certified
- Residual Solvents
- Within ICH limits
- Transport Information
- Not regulated for transport
- Lead Content
- Not more than 0.001%
- Boiling Point
- Not applicable (decomposes upon heating)
- Related Substances
- Not more than 0.5%
- Packing
- 25 kg fiber drum with double polyethylene bags inside
- Stability
- Stable under recommended storage conditions
- Loss on Drying
- Not more than 0.5%
Ethamsylate IP Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Payment Terms
- Cash in Advance (CID), Cash Advance (CA)
- Supply Ability
- 5000 Kilograms Per Day
- Delivery Time
- Days
- Sample Available
- Yes
- Sample Policy
- Sample costs shipping and taxes has to be paid by the buyer
- Packaging Details
- Fiber Drums
- Main Export Market(s)
- Australia, North America, Eastern Europe, Middle East, Africa, Western Europe, Central America, South America, Asia
- Main Domestic Market
- All India
- Certifications
- GMP,ISO
About Ethamsylate IP
Having core competency in the domain, we are able to provide the best grade Ethamsylate IP. Applauded for its excellent effectiveness, this intermediate is widely used for the management of blood loss in menorrhagia after surgery. Offered intermediate is processed using top-notch quality chemical compounds and innovative methodology by our dexterous professionals. Moreover, we provide this Ethamsylate IP in safe packaging options within prescribed time frame to our global patrons.
Features:
- Standard shelf life
- Safe to use
- Balanced composition
- Purity
FAQs:
Q: What is Ethamsylate?
A: Ethamsylate is a synthetic medication that is used to prevent or reduce bleeding in various medical conditions such as heavy menstrual bleeding, nosebleeds, and bleeding during or after surgery.
Q: How does Ethamsylate work?
A: Ethamsylate works by helping to stabilize blood vessels and prevent them from leaking blood. It also helps to increase platelet adhesiveness, which helps to stop bleeding.
Q: How is Ethamsylate taken?
A: Ethamsylate is typically taken orally in tablet form, usually 2-3 times per day. The dosage and duration of treatment will depend on the specific medical condition being treated.
Q: Are there any side effects associated with Ethamsylate?
A: Like all medications, Ethamsylate can cause side effects, although not everyone will experience them. Common side effects may include nausea, vomiting, headache, and dizziness. If you experience any severe side effects or allergic reactions, seek medical attention immediately.
Reliable Hemostatic Solution
Ethamsylate IP is trusted for its ability to stabilize capillary endothelium and effectively reduce bleeding episodes. With its pharmaceutical-grade purity exceeding 99%, this compound ensures consistent results for various formulations, supporting healthcare professionals in the treatment and prevention of hemorrhagic conditions. Its stability and odorless nature make it a preferred choice in pharmaceutical manufacturing.
Safe and Compliant Packaging
Packed in robust 25 kg fiber drums with double polyethylene bags inside, Ethamsylate IP is well-protected from moisture and light, preserving its quality during storage and transportation. The product is not classified as hazardous for transport, ensuring smooth logistics and compliance with safety standards for both distributors and manufacturers.
Quality Assurance and Compliance
Each batch of Ethamsylate IP meets the stringent requirements outlined by the Indian Pharmacopoeia, with GMP certification endorsing best manufacturing practices. Parameters like residual solvents, heavy metals, and loss on drying are rigorously controlled, reinforcing confidence in its application across pharmaceutical industries worldwide.
FAQs of Ethamsylate IP:
Q: How should Ethamsylate IP be stored to maintain its stability?
A: Ethamsylate IP should be kept in tightly closed containers at room temperature, protected from moisture and light to preserve its integrity over its 5-year shelf life.Q: What is the recommended usage of Ethamsylate IP in pharmaceutical manufacturing?
A: Ethamsylate IP serves as an active pharmaceutical ingredient for the formulation of tablets, capsules, and injectables used in the treatment and prevention of hemorrhage and capillary bleeding.Q: When is Ethamsylate IP typically applied in medical treatments?
A: Ethamsylate IP is employed during instances of hemorrhage, capillary bleeding, and to prevent postoperative bleeding, supporting faster restoration of normal vascular function.Q: Where can the structural formula of Ethamsylate IP be obtained?
A: The structural formula for Ethamsylate IP is available upon request from the manufacturer, supplier, or distributor.Q: What benefits does Ethamsylate IP offer in pharmaceutical formulations?
A: Ethamsylate IP promotes capillary stability and reduces bleeding time, making it a valuable component for effective hemostatic therapies in clinical settings.Q: Is there any regulatory concern for transporting Ethamsylate IP?
A: Ethamsylate IP is not regulated as hazardous material for transport, ensuring ease of logistics for distributors and exporters.Q: How is quality and safety assured for Ethamsylate IP?
A: Quality is validated through adherence to Indian Pharmacopoeia standards, GMP certification, and testing for heavy metals, residual solvents, and loss on drying.

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Ethamsylate Category
Ethamsylate UPS
Price 500 INR / Kilograms
Minimum Order Quantity : 100 Kilograms
Physical Form : Powder
Storage : Other, Store in a cool, dry place away from light and moisture
Ph Level : Neutral (6.57.5)
Taste : Other, Slightly bitter
Ethamsylate BP
Price 500 INR / Kilograms
Minimum Order Quantity : 100 Kilograms
Physical Form : Powder
Storage : Other, Store in a cool, dry place, protected from light
Ph Level : 5.57.5 (1% solution)
Taste : Other, Practically tasteless

 Send Inquiry
Send Inquiry


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese